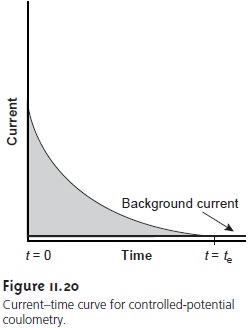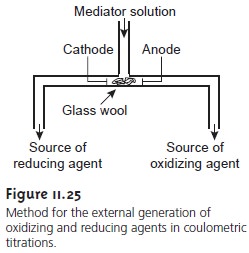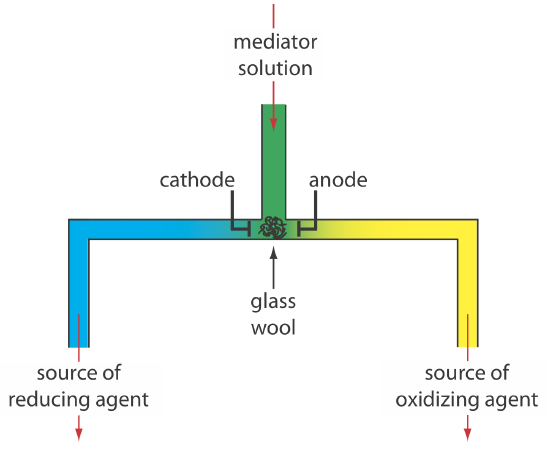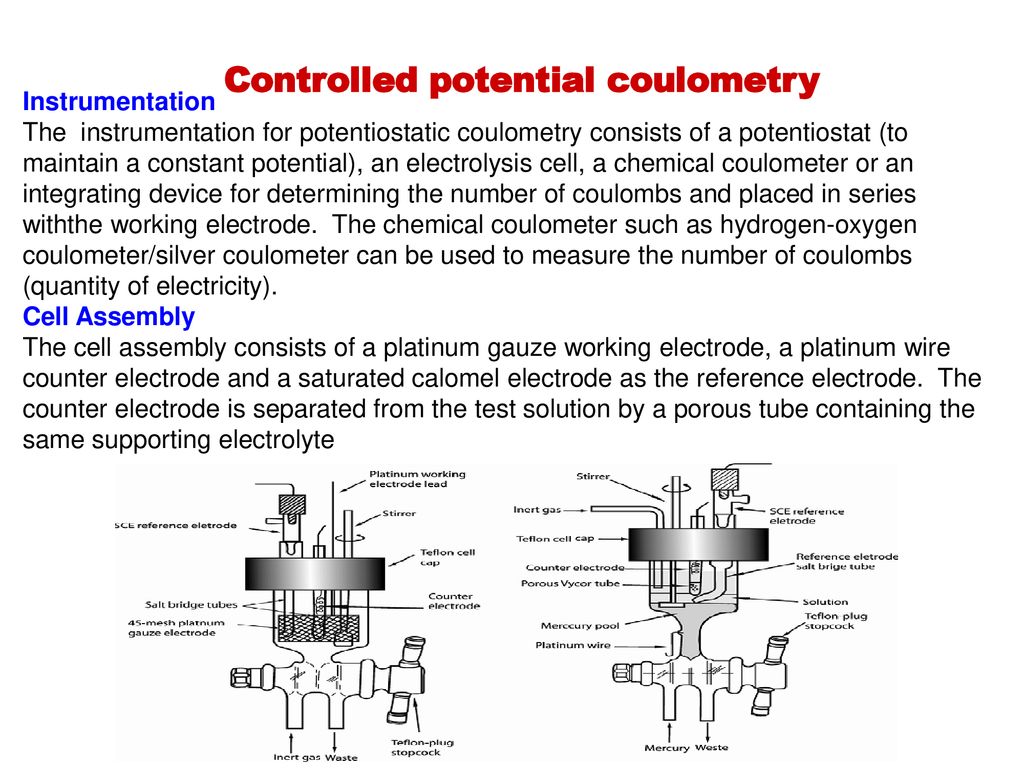
A.) Introduction : 1.) Coulometry: electrochemical method based on the quantitative oxidation or reduction of analyte - measure amount of analyte by measuring. - ppt download

Sensors | Free Full-Text | Coulometer from a Digitally Controlled Galvanostat with Photometric Endpoint Detection

Sensors | Free Full-Text | Determination of Trace Amounts of Hydrofluoric Acid in Non-Aqueous Solutions by the Coulometric Titration Method