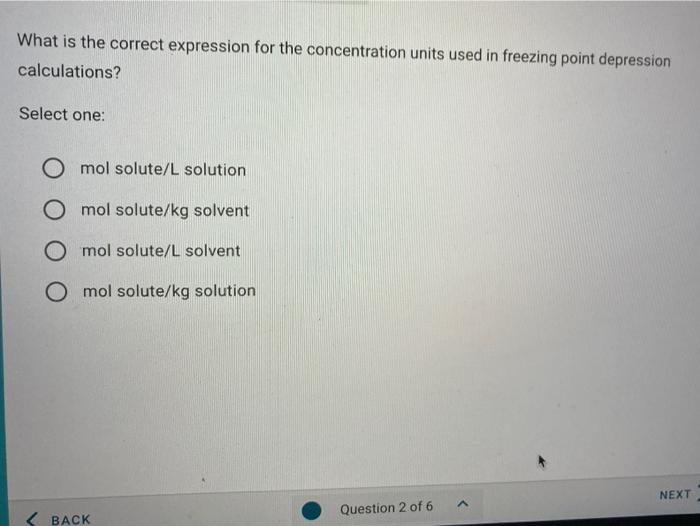
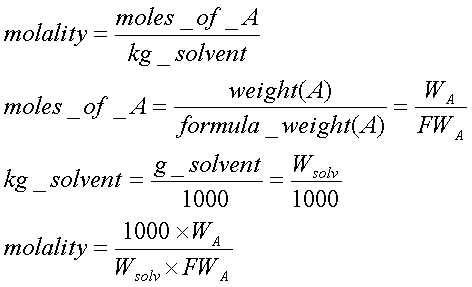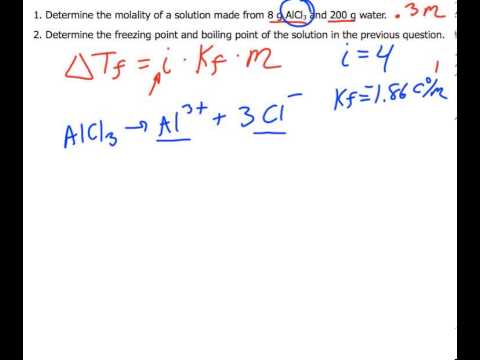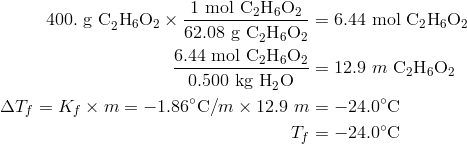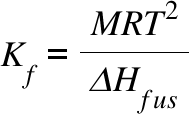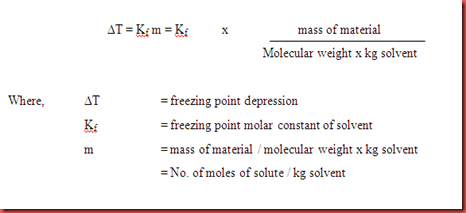A brief introduction to freezing point depression THE COLLIGATIVE PROPERTIES OF MOLALITY AND FREEZING POINT DEPRESSION grownextgen.org. - ppt download
calculate molal depression cons†an t of solvent , which has freezing point 16.6^0 celsius, andblatent heat of fusion 180.7

How do you find the freezing point of pure water from the freezing point depression equation? | Homework.Study.com

Calculate the molal depression constant of a solvent which has freezing point 16.6^(@)C and latent heat of fusion 180.75 "J g"^(-1) :

Colligative Properties - Boiling Point Elevation, Freezing Point Depression & Osmotic Pressure - YouTube
Calculate the depression in the freezing point of water when 10 g of CH3CH2CHClCOOH is - Sarthaks eConnect | Largest Online Education Community

10: Determination of the Molar Mass by Freezing Point Depression (Experiment) - Chemistry LibreTexts

Calculate the freezing point and the boiling point at 1 atmosphere of a solution containing 30 g cane sugar (molecular mass 342 ) and 150 g water.Given : Kb = 0.513 and Kf = 1.86