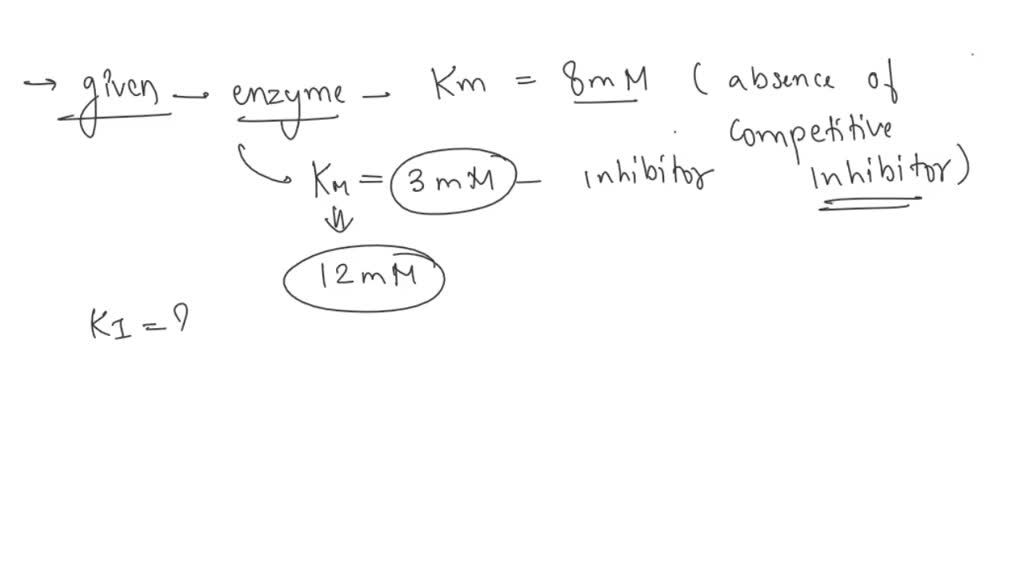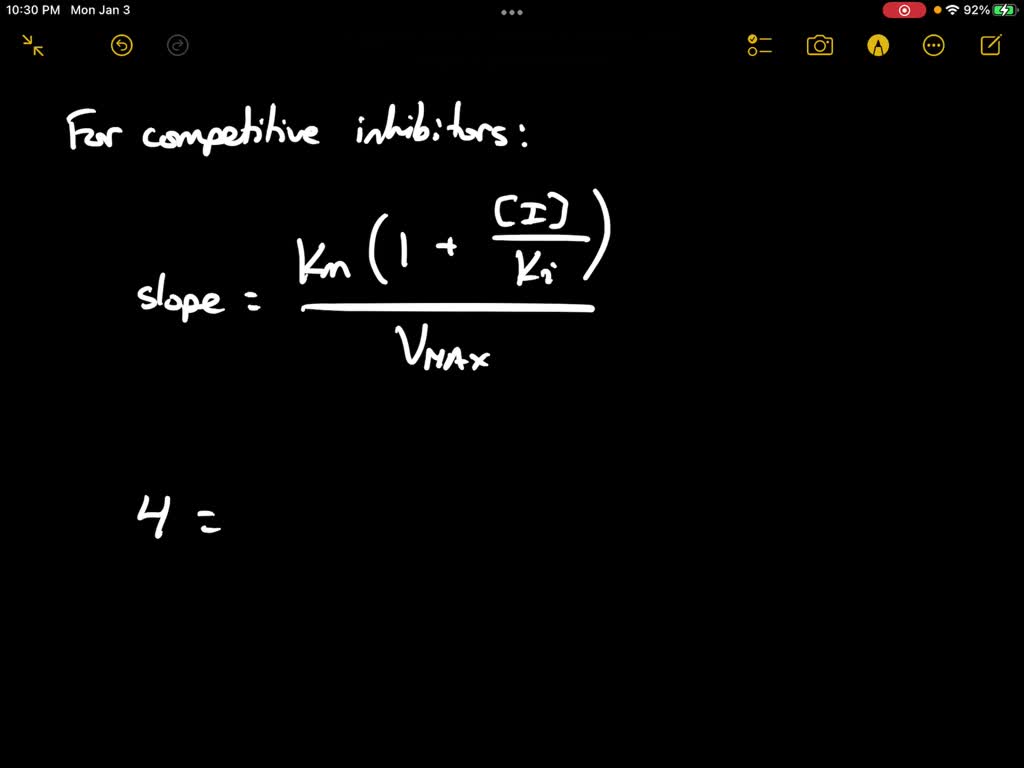
SOLVED: Calculate the Ki for a competitive inhibitor whose concentration = 200 mg/mL, Km = 0.80, vmax = 0.20, slope = 4. Please show work.

Lineweaver-Burk secondary plot for Ki calculation activity. Compound 3b... | Download Scientific Diagram

LAB 3 Enzyme Kinetics Studying -galactosidase activity at varying substrate concentrations in the presence and absence of an inhibitor Michaelis-Menten. - ppt video online download

Calculate KC for the reaction, KI + I2 KI3 . Given that initial weight of KI is 1.326 g . Weight of KI3 is 0.105 g and no. of mole of free









![Solved 1/v vs. 1/Ipyruvate] without inhibitor 90 80 60 50 40 | Chegg.com Solved 1/v vs. 1/Ipyruvate] without inhibitor 90 80 60 50 40 | Chegg.com](https://d2vlcm61l7u1fs.cloudfront.net/media%2F8ed%2F8ed73308-e0da-482e-9248-19bdd0cab0ae%2FphpycFrAI.png)