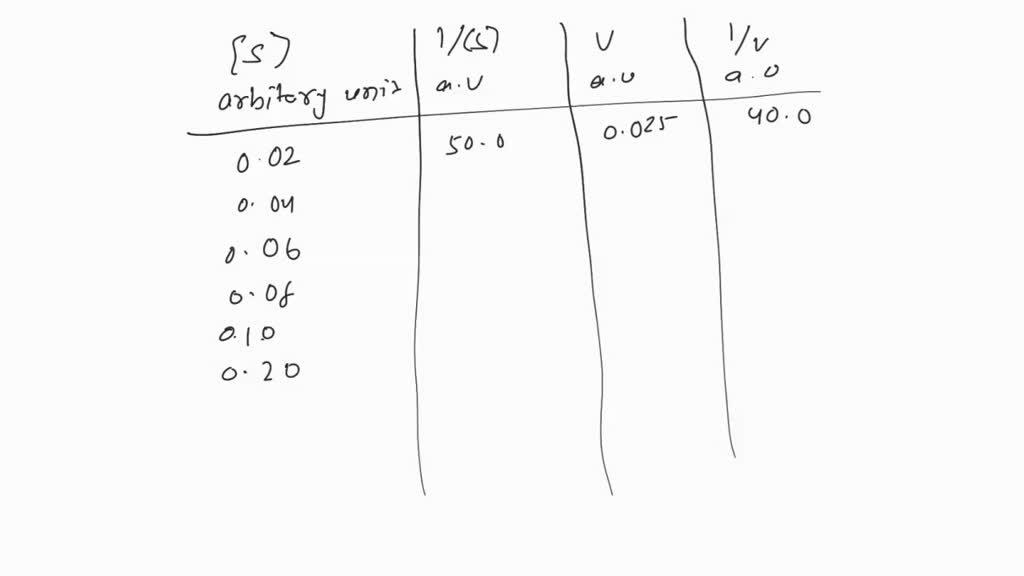
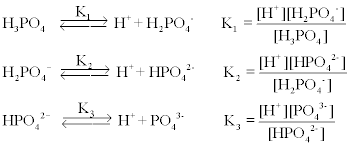Preparation of Buffers - 1 Calculate the volume of sulfuric acid (H 2 SO 4 ) necessary to prepare 600 milliliter 0.5M H 2 SO 4 from concentrated H 2 SO. - ppt download

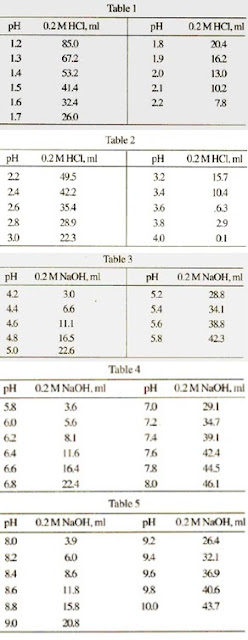
Table 1 from Dynamic approach to predict pH profiles of biologically relevant buffers | Semantic Scholar

SOLVED: 8 Consider beaker containing equal amounts of dihydrogen potassium phosphate (monobasic) and potassium hydrogen phosphate (dibasic) to which either a strong acid or strong base is added:. Calculate the pH of